Liquid biopsy is a simple blood test that detects cancer DNA circulating in the bloodstream, allowing doctors to find, monitor, and track cancer without any surgery.
In 2026, it has become one of the most important tools in oncology: new AI-powered tests can detect brain tumours at 98% accuracy from a blood draw alone, multi-cancer screening platforms can flag 50+ cancer types simultaneously, and a landmark U.S. law now guarantees Medicare coverage for these tests starting in 2029.
For the first time, catching cancer early before symptoms appear is becoming a realistic possibility for millions of people worldwide.
Key Takeaways
- Blood is becoming the new biopsy – In 2026, liquid biopsy can detect cancer earlier, monitor treatment in real time, and catch relapse up to 13 months before it shows on a scan.
- AI is unlocking cancers once beyond liquid biopsy’s reach – New AI + nanosensor technology detects brain tumours at 98% accuracy, and the M-PACT algorithm classifies pediatric brain tumours at 92% accuracy from spinal fluid.
- The cost-sensitivity barrier is finally breaking – The Bridge Capture sequencing method delivers high sensitivity at 10x lower sequencing depth, making accurate, affordable liquid biopsy realistic for community clinics and lower-resource settings.
- Challenges remain, but the direction is clear – Early-stage sensitivity, false positives from clonal hematopoiesis, and health equity gaps are real obstacles, but every major 2026 breakthrough has pushed these boundaries further, and the momentum is accelerating.
What Is Liquid Biopsy?
At its core, a liquid biopsy is any diagnostic test performed on a bodily fluid, most commonly blood, but also urine, cerebrospinal fluid, or saliva, to detect signs of cancer without physically removing tumour tissue.
When cancer cells grow, divide, or die, they release biological material into the bloodstream. The most studied of these is circulating tumour DNA (ctDNA), fragments of DNA carrying the tumour’s unique genetic fingerprint. But liquid biopsies in 2026 are increasingly multimodal, analysing a rich combination of signals:
- ctDNA – Tumour DNA fragments carrying mutations, methylation patterns, and chromosomal rearrangements
- Circulating Tumour Cells (CTCs) – Intact cancer cells shed directly into the blood
- Exosomes & Extracellular Vesicles – Tiny membrane bubbles released by tumours, packed with proteins and RNA
- Cell-Free RNA (cfRNA) – RNA signals that can indicate gene expression patterns in tumours
- Fragmentomics – The study of how tumour DNA breaks apart differently from healthy DNA, adding another diagnostic layer
- Tumour-Educated Platelets (TEPs) – Blood platelets that absorb tumour-derived molecules and carry detectable cancer signatures
When these signals are fed into machine learning models trained on millions of samples, the result is a diagnostic picture that no single tissue biopsy can match.
How It Differs from Traditional Biopsy
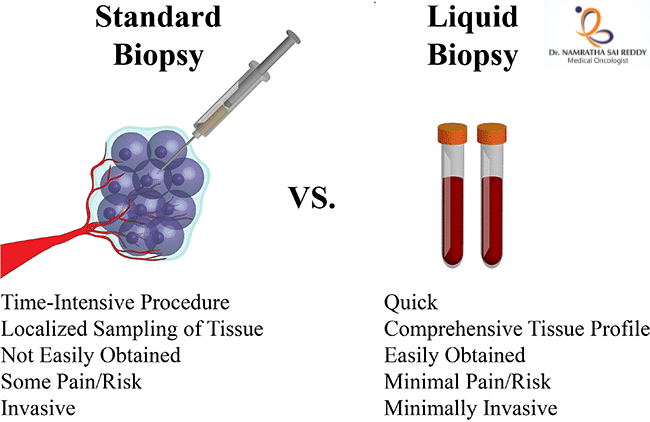
| Feature | Traditional Tissue Biopsy | Liquid Biopsy |
| Invasiveness | Surgical procedure required | Simple blood or fluid draw |
| Tumor representation | Single tumour region sampled | Captures signals from tumours throughout the body |
| Turnaround time | Days to weeks | Often 24–72 hours |
| Repeatability | Limited, risky to repeat often | Easily repeated at regular intervals |
| Real-time monitoring | Not feasible | Ideal for ongoing treatment monitoring |
| Early detection potential | Low, requires a visible tumour | High, detects ctDNA before symptoms appear |
| Risk to the patient | Bleeding, infection, procedural risk | Minimal — equivalent to a blood test |
| Tumor heterogeneity | Misses mutations in other regions | Samples DNA from across the tumour landscape |
| Cost (2026) | $1,500–$5,000+ | $300–$1,500 depending on test type |
The two approaches are most powerful when used together, tissue biopsy for initial diagnosis and molecular characterisation, liquid biopsy for monitoring, treatment selection, and early recurrence detection. The goal is not replacement but integration.
The Science: Why ctDNA Is Such a Powerful Signal
Every tumour is genetically distinct. As cancer cells proliferate and die, they leave behind fragments of DNA in the bloodstream, some as short as 160 base pairs. These fragments carry mutations specific to the tumour: point mutations, insertions, deletions, copy number variations, and methylation changes. Detecting them requires extreme precision, because in early-stage cancer, ctDNA can represent as little as 0.01% of all circulating DNA in the blood.
To isolate this signal from the noise, modern liquid biopsy platforms use:
- Next-Generation Sequencing (NGS) – Massively parallel sequencing that reads millions of DNA fragments simultaneously
- Digital PCR (ddPCR) – Droplet-based amplification that can detect one mutant molecule in 100,000 normal ones
- Methylation profiling – Analyses chemical tags on DNA that indicate which tissue a fragment originated from, enabling cancer-of-origin determination
- AI and machine learning – Trained models that integrate multiple signal types to distinguish true cancer signals from background noise, ageing-related mutations, and clonal hematopoiesis artefacts
The sensitivity challenge remains the field’s central technical problem, particularly for stage I cancers where tumour shedding is minimal. But every major advance in 2026 has pushed this boundary further.
The Major Breakthroughs of Early 2026
1. AI + Nanosensors Solve the Brain Tumour Problem
Brain tumours have historically been liquid biopsies’ most stubborn challenge. The blood-brain barrier – the tight cellular seal that protects the brain from pathogens — also blocks most tumour DNA from leaking into the bloodstream, making standard ctDNA detection unreliable for gliomas and other CNS cancers.
In January 2026, Memorial Sloan Kettering Cancer Centre announced a landmark solution. Their team developed a liquid biopsy platform combining carbon nanotube sensors with artificial intelligence that detects and classifies brain tumours with 98% accuracy.
Rather than searching for tumour DNA in the blood, the nanosensors detect subtle systemic changes, shifts in immune proteins, metabolic signals, and inflammatory markers throughout the body triggered by the presence of a brain tumour. Researchers describe this as reading the tumour’s “ecosystem effect” on the entire body, rather than trying to catch the few DNA fragments that escape the blood-brain barrier.
Why it matters:
- Brain tumour patients currently require craniotomy (open brain surgery) or stereotactic needle biopsy just to get a tissue diagnosis — both carry serious neurological risk
- This test requires nothing more than a standard blood draw
- It can classify tumour type and grade, critical for treatment planning
- Early detection of glioblastoma, the most aggressive form, could allow surgery and treatment to begin while the tumour is smaller and more operable
2. M-PACT: AI Classifies Pediatric Brain Tumours from Spinal Fluid
Published in Nature Cancer in February 2026, St. Jude Children’s Research Hospital’s M-PACT (Methylation-based Predictive Algorithm for CNS Tumours) represents a parallel revolution for pediatric oncology. The algorithm analyses ctDNA extracted from cerebrospinal fluid (CSF) and uses DNA methylation patterns to classify brain tumour subtypes with 92% accuracy.
Key capabilities of M-PACT include:
- Tumour subtype classification – Distinguishes between medulloblastoma, ependymoma, diffuse midline glioma, and dozens of other pediatric CNS tumours based on methylation signature alone
- Relapse vs. new tumour distinction – Can tell whether a new lesion represents a recurrence of the original cancer or an entirely separate second tumour, a distinction that fundamentally changes whether the child needs salvage chemotherapy or a new primary treatment protocol
- Minimal tissue requirement – Works on the small ctDNA fragments found in CSF, avoiding the need for repeat surgical biopsies in children
For context, pediatric brain tumours are the leading cause of cancer-related death in children under 15. The ability to non-invasively monitor these children throughout treatment and catch relapse earlier could have a direct impact on survival rates.
3. Bridge Capture: Finally Solving the Speed-Sensitivity-Cost Triangle
For years, liquid biopsy developers faced an iron triangle: you could have a test that was sensitive, fast, or cheap, but rarely all three simultaneously. High sensitivity required deep sequencing, which was slow and expensive. Simplified workflows sacrificed rare-mutation detection.
In January 2026, researchers from Genomill Health, the University of Turku, and Turku University Hospital published results on Bridge Capture, a targeted next-generation sequencing enrichment method designed to break this tradeoff. Key findings:
- Demonstrated superior sensitivity for rare ctDNA variants compared to existing targeted sequencing methods
- Achieved consistent performance even at 10x lower sequencing depth, dramatically reducing cost per sample
- Produced equivalent results across manual and automated laboratory workflows, enabling standardised clinical deployment
- Validated across multiple clinical centres, confirming real-world reproducibility
Bridge Capture is particularly significant not because it is the most sensitive test ever created, but because it may be the first to simultaneously meet the sensitivity, cost, throughput, and standardisation requirements needed for population-scale cancer screening.
4. Lung Cancer: Detecting Mutations Six Months Before Diagnosis
Research published in Molecular Oncology in early 2026 from Hospital de Amor de Barretos in Brazil delivered one of the year’s most striking findings: in an asymptomatic study participant, a liquid biopsy identified a TP53 gene mutation six months before their lung cancer diagnosis.
The study evaluated commercial multigene liquid biopsy panels in non-small cell lung cancer patients and found:
- ctDNA panels successfully identified actionable mutations without specialised collection protocols
- Detection was possible at earlier stages than standard imaging
- The findings confirm the feasibility of liquid biopsy as a lung cancer screening tool for high-risk populations, smokers, those with occupational exposures, and individuals with family history
- No special blood processing equipment was required, making the approach deployable in standard clinical labs
Lung cancer kills more people globally than any other cancer, largely because most cases are caught at stage III or IV when treatment options are limited. The ability to detect it months before symptoms appear could shift the diagnosis distribution dramatically toward earlier, more treatable stages.
Key Statistics at a Glance (2026)
| Metric | Figure |
| Brain tumour detection accuracy (AI + nanosensor) | 98% |
| Pediatric brain tumour classification accuracy (M-PACT) | 92% |
| Relapse detection lead time vs. imaging | 6–13 months |
| ctDNA detected before lung cancer diagnosis | 6 months |
| Guardant360 CDx FDA-approved indications | 6 approvals |
| Medicare MCED coverage start year (Miller-Meeks) | 2029 |
| Approximate MCED test cost under Medicare | $500 |
Regulatory and Coverage Milestones
2026 has been as consequential in the boardroom and legislature as in the lab. Several pivotal regulatory and coverage events have reshaped the liquid biopsy landscape:
The Miller-Meeks Bill (Signed into Law, January 2026). Perhaps the single most impactful development for patient access: the Medicare Multikancer Early Detection Screening Coverage Act, commonly called the Miller-Meeks Bill, was signed into law. Key provisions:
- FDA-approved MCED tests become eligible for Medicare coverage beginning in 2029
- Initial coverage applies to Americans aged 50 to 65
- Estimated cost to Medicare per test: approximately $500
- Coverage expands the reach of early cancer detection to an estimated 60+ million Medicare beneficiaries over time
Where Liquid Biopsy Is Being Used Today
Liquid biopsy has moved well beyond experimental trials. In 2026, it is actively deployed across the cancer care continuum:
- Treatment selection – Identifying actionable mutations (EGFR, ALK, BRAF, KRAS, HER2) in lung, colorectal, and breast cancers to match patients with targeted therapies or immunotherapy
- Minimal residual disease (MRD) monitoring – Detecting microscopic residual cancer after surgery or chemotherapy that predicts future relapse
- Relapse surveillance – Serial blood draws every 3–6 months to catch recurrence before imaging
- Therapy resistance monitoring – Tracking the emergence of new mutations that signal a cancer is becoming resistant to current treatment, allowing earlier treatment switches
- Multi-cancer early detection – Annual blood-based screening for cancer signal across 50+ cancer types in asymptomatic individuals
- Clinical trial enrollment – Using liquid biopsy to identify patients with specific mutations who qualify for targeted therapy trials
The Challenges That Still Remain
Despite the remarkable momentum, the field is candid about its unsolved problems:
- Early-stage sensitivity – ctDNA is extremely scarce in stage I cancers. Many tests still miss 30–50% of early-stage cases, which is precisely when detection matters most
- Tissue-of-origin localisation – Some MCED tests detect a cancer signal but cannot yet pinpoint with certainty which organ it originated from, leading to extensive (and sometimes unnecessary) follow-up imaging
- False positives – Clonal hematopoiesis, age-related mutations in blood stem cells can mimic cancer signals, generating false alarms that cause patient anxiety and unnecessary procedures
- Standardisation – No universal standards exist yet for ctDNA reporting, variant calling thresholds, or clinical interpretation, making cross-platform comparison difficult
- Health equity – The benefits of liquid biopsy are currently concentrated in high-income countries and large academic medical centres. Bringing these tools to community clinics, rural settings, and low-and-middle-income countries remains an urgent, unfinished task
- Clinical action pathways – Detecting a cancer signal is only the first step. Clear, validated follow-up protocols for every cancer type and stage are still being developed
The Outlook: Blood as the New Biopsy
The trajectory of liquid biopsy in 2026 is unmistakable. Brain tumours, once the field’s most resistant frontier, are falling to AI-powered nanosensors. Children with brain cancer can be monitored through spinal fluid rather than repeated surgeries. A sequencing method has finally cracked the speed-sensitivity-cost triangle. And for the first time, a U.S. law guarantees that MCED screening will be accessible to tens of millions of Medicare beneficiaries.
These are not isolated breakthroughs. They are the compounding returns of two decades of investment in genomics, machine learning, nanotechnology, and clinical validation, all converging at once.
The vision that animates this entire field is simple and profound: cancer should be caught in the blood, not in the emergency room. Every day, that vision gets closer to being routine. One blood draw at a time.
FAQ
1. What is a liquid biopsy?
A blood test that detects cancer DNA circulating in the bloodstream, no surgery required.
2. What is a liquid biopsy used for?
Early cancer detection, treatment selection, therapy monitoring, and catching relapse — often months before symptoms or scans reveal anything.
3. How accurate is it in 2026?
AI-powered tests now detect brain tumours at 98% accuracy. Multi-cancer platforms screen for 50+ cancer types from a single blood draw.
4. How early can it detect cancer?
Up to 6 months before diagnosis in some cases, and relapse up to 13 months before it appears on imaging.
5. Is it different from a regular biopsy?
Yes. A tissue biopsy requires surgery. Liquid biopsy is a simple blood draw, faster, safer, and repeatable.
6. Which cancers can it detect?
Lung, colorectal, breast, bladder, brain, and 50+ others via multi-cancer early detection platforms like Galleri.
7. Is it covered by insurance?
Companion diagnostics tied to specific treatments are often covered. Medicare coverage for MCED tests begins in 2029 under the Miller-Meeks Bill.
8. What is ctDNA?
Circulating tumour DNA, fragments of cancer cell DNA shed into the bloodstream that liquid biopsy tests detect and analyse.