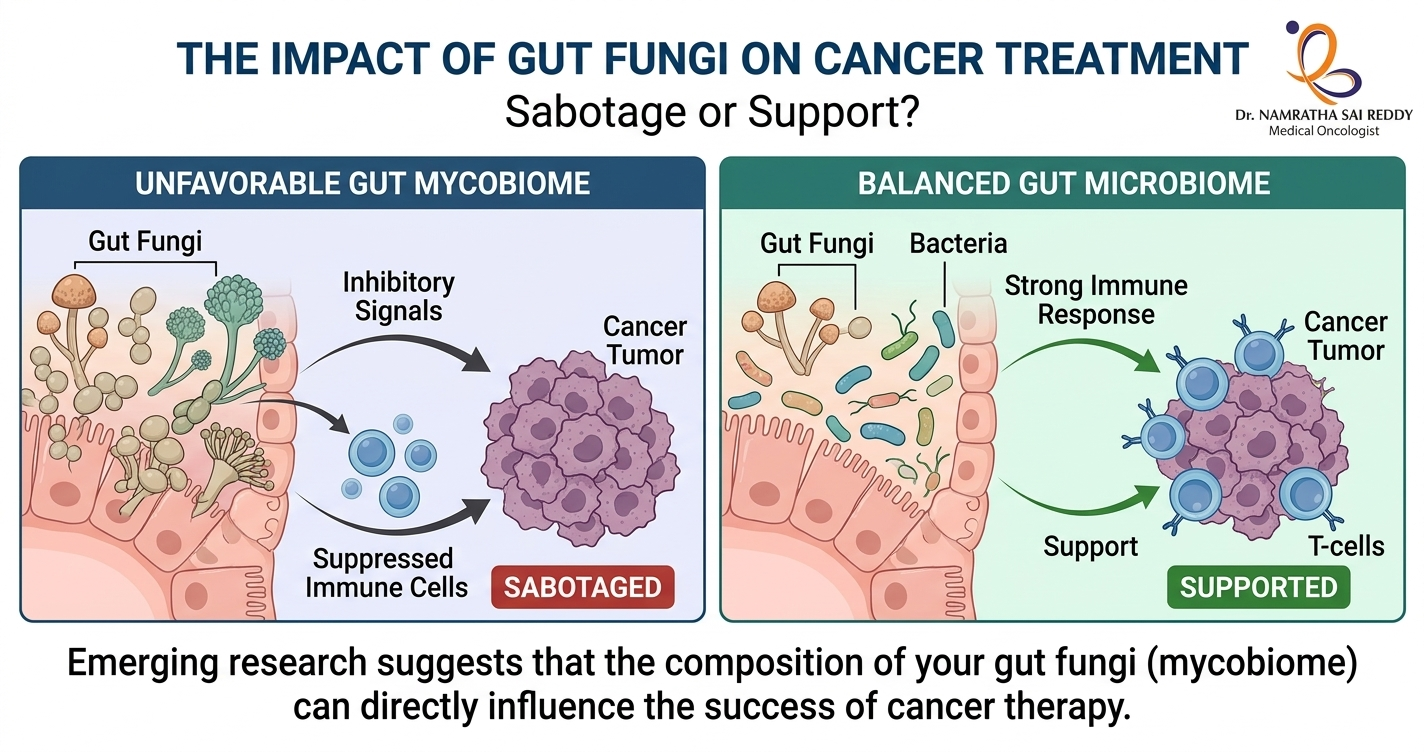
Yes, gut fungi can affect cancer treatment outcomes. Research published in Cancer Cell by scientists at Cedars-Sinai Medical Centre found that intestinal fungi suppress anti-tumour immune responses after radiation therapy. Fungal overgrowth, which frequently follows antibiotic use, was shown to reduce treatment effectiveness in models of both breast cancer and melanoma. Treating the animals with antifungal drugs restored the immune response and improved outcomes. Scientists now believe the gut mycobiome may be one of the missing pieces explaining why only 20 to 40% of patients respond to immunotherapy.
Key Takeaways
- Gut fungi (the mycobiome) can suppress the immune system’s ability to fight cancer after radiation therapy and immunotherapy.
- Antibiotic use can trigger fungal overgrowth in the gut, potentially reducing cancer treatment effectiveness.
- A fungal-sensing immune receptor called Dectin-1 has been linked to worse survival in breast cancer and melanoma patients with elevated tumour expression.
- Specific fungi such as Candida albicans and Malassezia are associated with colorectal, liver, and pancreatic cancer progression.
- Microbiome profiling and targeted antifungal strategies are being actively investigated as ways to improve immunotherapy response rates.
What Is the Gut Mycobiome?
The human gut hosts approximately 38 trillion microorganisms, including bacteria, fungi, viruses, and other microbes. While research has traditionally focused on bacteria, the gut’s fungal community (the mycobiome) is now attracting serious scientific attention.
Fungi make up just 0.01 to 2% of cells in the gut microbiome, but researchers have reliably identified 50 to 100 fungal genera living there. These fungi colonise early in life through birth, breastfeeding, food intake, and environmental exposure. Under normal conditions, they are non-pathogenic. However, when bacterial populations decline or the immune system is disrupted, fungal communities can shift dramatically, with serious downstream effects on immunity and treatment response.
The Cedars-Sinai Study: Fungi and Radiation Therapy
The most significant study in this field to date was led by Dr. Stephen Shiao and Dr. David Underhill at Cedars-Sinai Medical Centre. Published in Cancer Cell, the research found that intestinal bacteria and fungi have directly opposing effects on anti-tumour immune responses following radiation therapy. Consistent results were observed across both breast cancer and melanoma models, suggesting the findings may apply broadly across cancer types.
Key findings:
- When gut bacteria were reduced using antibiotics, fungi rapidly expanded in the gut.
- This fungal overgrowth weakened the immune system’s anti-tumour response after radiation therapy.
- Treating the animals with antifungal drugs restored immune activity and improved radiation therapy effectiveness.
- Germ-free mice with no fungi showed enhanced responses to radiation therapy compared to mice with normal fungal populations.
This is clinically relevant because antibiotics are routinely prescribed to cancer patients who are at elevated infection risk. The study suggests that this common practice may, in some cases, inadvertently compromise treatment outcomes by promoting fungal overgrowth.
The Dectin-1 Connection: How Tumours Sense Fungi
A key discovery from the Cedars-Sinai research was the role of a fungal-sensing immune receptor called Dectin-1. This protein detects beta-1,3-glucan, a compound in fungal cell walls, and activates the body’s antifungal immune pathways. It is primarily expressed in macrophages and dendritic cells within tumours.
Using data from The Cancer Genome Atlas, the researchers found:
- Breast cancer patients with above-average Dectin-1 expression in their tumours had worse survival outcomes than those with lower levels.
- Similar results were observed in melanoma patients.
- Mice engineered to lack Dectin-1 showed enhanced responses to radiation therapy, confirming the receptor’s role in suppressing anti-tumour immunity.
As the National Cancer Institute noted in its coverage of the study, Dectin-1 could serve as a biomarker to identify patients with an unfavourable microbiome composition ahead of treatment.
Specific Fungi Linked to Cancer Risk
A comprehensive review published in Gut Microbes (2025) highlights specific fungal species now linked to distinct cancer outcomes:
- Candida albicans: Overgrowth has been linked to colorectal and liver cancer progression.
- Malassezia: Expansion of this species contributes to pancreatic carcinogenesis through complement activation.
The review also notes that in cancer patients, the gut mycobiome undergoes substantial remodelling in both its composition and ecological structure. These shifts are increasingly recognised as important modulators of disease progression, immune dynamics, and responsiveness to therapy.
How Gut Fungi Influence Immunotherapy
Immunotherapy works by activating immune cells to recognise and destroy cancer. Research published in npj Biofilms and Microbiomes (2025) outlines the key mechanisms through which gut fungi influence this process.
Immune signalling modulation – Fungal molecules interact with immune receptors such as Dectin-1 and can shift the immune environment towards tolerance and suppression rather than active tumour-fighting.
Tumour microenvironment changes – According to a 2026 review in Gut Microbes, fungi and their metabolites remodel the tumour microenvironment through metabolic reprogramming, including depletion of critical amino acids and production of immunosuppressive short-chain fatty acids.
Microbial balance disruption – When bacterial diversity drops following antibiotic use, poor diet, or illness, fungi overgrow and disrupt the anti-tumour immune activity that bacteria normally support. This balance is now considered a critical regulator of cancer treatment response.
Impaired CD8 T cell activity – The Cedars-Sinai study found that fungal overgrowth impairs the activity of cytotoxic CD8 T cells (the immune cells most critical for killing cancer cells) while increasing immunosuppressive macrophages in the tumour microenvironment.
A Fast-Moving Field: Research Milestones
The relationship between the gut microbiome and immunotherapy is one of oncology’s fastest-growing research areas. Research published in iMeta (2024) documents the following milestones:
- 2015: Scientists first demonstrated a direct link between gut bacteria and immune checkpoint inhibitor response.
- 2018: Clinical studies explored gut microbial metabolites as predictive markers for ICI effectiveness.
- 2020 to 2021: Clinical breakthroughs demonstrated that faecal microbiota transplantation could enhance ICI response in immunotherapy-resistant melanoma patients.
- 2022 to 2023: Studies correlated gut microbiome composition with CAR-T cell therapy efficacy using next-generation sequencing and machine learning.
- 2024 to 2025: Research focus shifted to multi-omics and metabolic mechanisms, including the previously underappreciated role of fungi.
A landmark review in Nature Reviews Drug Discovery (2025) concludes that the gut microbiome is now recognised as a key modulator of immunotherapy efficacy, with clinical trials of microbiome-based interventions actively underway.
The Future of Microbiome-Driven Cancer Care
Researchers highlighted in a Nature collection on the microbiome in cancer (2024) note that tumours contain both bacteria and fungi, and that their presence is proving useful in detecting cancers, categorising them, and predicting whether specific interventions will work.
Potential future strategies include:
- Mycobiome profiling before treatment to predict response to immunotherapy and radiation therapy
- Targeted microbiome modulation through diet, probiotics, or antifungal therapies given alongside cancer treatment
- Faecal microbiota transplantation to improve treatment response in immunotherapy-resistant patients
- Dectin-1 and other fungal-sensing proteins as biomarkers for treatment stratification
- Precision oncology approaches that integrate both bacterial and fungal microbiome health with standard cancer treatment planning
A 2026 review in Gut Microbes concludes that fungi and their metabolites may enhance tumour immunogenicity and reduce therapeutic resistance, with antifungal-based therapies showing particular promise as an adjunct to immunotherapy.
What This Means for Cancer Patients
Cancer therapy is no longer just about targeting tumour cells. The microbial ecosystem inside the body plays a significant role in determining treatment success.
- The gut mycobiome actively influences immune activity in ways relevant to cancer treatment.
- Fungal imbalance, particularly following antibiotic use, may weaken responses to radiation and immunotherapy.
- Specific fungal species and immune markers like Dectin-1 may serve as future biomarkers for personalised treatment planning.
- Future cancer care may include routine microbiome profiling and targeted microbiome management as part of standard treatment protocols.
As Dr. David Underhill of Cedars-Sinai stated after the landmark study: “It would be really handy to be able to characterise a patient’s microbiome, both bacterial and fungal, and determine their expected prognosis, or how they will respond to radiation therapy, chemotherapy, or immunotherapy. Then we would like to see whether it is feasible to change that microbiome to make those therapies work better.”
The gut microbiome, including its fungal residents, may soon become one of oncology’s most important biomarkers and therapeutic targets.
FAQs
Can gut fungi actually affect cancer treatment outcomes?
Yes. Research in Cancer Cell shows that gut fungi directly suppress anti-tumour immune responses after radiation therapy. Antifungal treatment restored immune activity and improved outcomes in preclinical models of breast cancer and melanoma.
Why do some patients not respond to immunotherapy?
Multiple factors contribute, including tumour genetics, immune profile, and gut microbiome composition. Only 20 to 40% of patients in certain groups respond to immunotherapy. Scientists believe optimising the mycobiome may help improve these rates.
What is Dectin-1, and why does it matter in cancer?
Dectin-1 is an immune receptor that senses fungi. Higher Dectin-1 expression in tumours has been linked to worse survival in breast cancer and melanoma patients. It is being studied as a potential biomarker for predicting treatment response.
What is the gut mycobiome?
The gut mycobiome is the community of fungi living in the intestinal tract. An imbalance, called fungal dysbiosis, is linked to poorer cancer outcomes and reduced treatment response.
Can diet affect the gut mycobiome and cancer treatment?
Emerging research suggests diet, probiotics, and antifungal interventions can all influence the mycobiome. High-sugar diets promote Candida growth. Specific dietary recommendations for cancer patients are still under investigation.