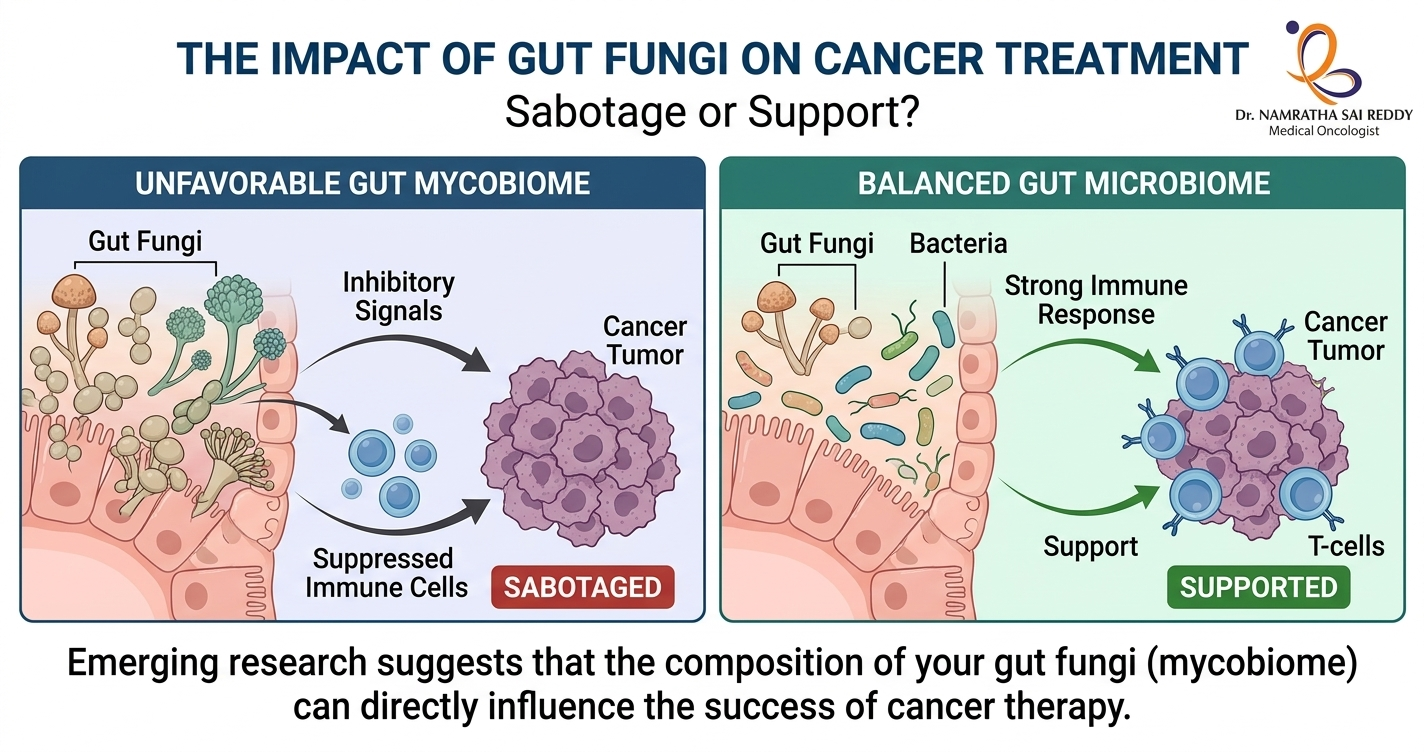
Yes, gut fungi can affect cancer treatment outcomes. Research published in Cancer Cell by scientists at Cedars-Sinai Medical Centre found that intestinal fungi suppress anti-tumour immune responses after radiation therapy. Fungal overgrowth, which frequently follows antibiotic use, was shown to reduce treatment effectiveness in models of both breast cancer and melanoma. Treating the animals with antifungal drugs restored the immune response and improved outcomes. Scientists now believe the gut mycobiome may be one of the missing pieces explaining why only 20 to 40% of patients respond to immunotherapy. Key Takeaways What Is the Gut Mycobiome? The human gut hosts approximately 38 trillion microorganisms, including bacteria, fungi, viruses, and other microbes. While research has traditionally focused on bacteria, the gut’s fungal community (the mycobiome) is now attracting serious scientific attention. Fungi make up just 0.01 to 2% of cells in the gut microbiome, but researchers have reliably identified 50 to 100 fungal genera living there. These fungi colonise early in life through birth, breastfeeding, food intake, and environmental exposure. Under normal conditions, they are non-pathogenic. However, when bacterial populations decline or the immune system is disrupted, fungal communities can shift dramatically, with serious downstream effects on immunity and treatment response. The Cedars-Sinai Study: Fungi and Radiation Therapy The most significant study in this field to date was led by Dr. Stephen Shiao and Dr. David Underhill at Cedars-Sinai Medical Centre. Published in Cancer Cell, the research found that intestinal bacteria and fungi have directly opposing effects on anti-tumour immune responses following radiation therapy. Consistent results were observed across both breast cancer and melanoma models, suggesting the findings may apply broadly across cancer types. Key findings: This is clinically relevant because antibiotics are routinely prescribed to cancer patients who are at elevated infection risk. The study suggests that this common practice may, in some cases, inadvertently compromise treatment outcomes by promoting fungal overgrowth. The Dectin-1 Connection: How Tumours Sense Fungi A key discovery from the Cedars-Sinai research was the role of a fungal-sensing immune receptor called Dectin-1. This protein detects beta-1,3-glucan, a compound in fungal cell walls, and activates the body’s antifungal immune pathways. It is primarily expressed in macrophages and dendritic cells within tumours. Using data from The Cancer Genome Atlas, the researchers found: As the National Cancer Institute noted in its coverage of the study, Dectin-1 could serve as a biomarker to identify patients with an unfavourable microbiome composition ahead of treatment. Specific Fungi Linked to Cancer Risk A comprehensive review published in Gut Microbes (2025) highlights specific fungal species now linked to distinct cancer outcomes: The review also notes that in cancer patients, the gut mycobiome undergoes substantial remodelling in both its composition and ecological structure. These shifts are increasingly recognised as important modulators of disease progression, immune dynamics, and responsiveness to therapy. How Gut Fungi Influence Immunotherapy Immunotherapy works by activating immune cells to recognise and destroy cancer. Research published in npj Biofilms and Microbiomes (2025) outlines the key mechanisms through which gut fungi influence this process. Immune signalling modulation – Fungal molecules interact with immune receptors such as Dectin-1 and can shift the immune environment towards tolerance and suppression rather than active tumour-fighting. Tumour microenvironment changes – According to a 2026 review in Gut Microbes, fungi and their metabolites remodel the tumour microenvironment through metabolic reprogramming, including depletion of critical amino acids and production of immunosuppressive short-chain fatty acids. Microbial balance disruption – When bacterial diversity drops following antibiotic use, poor diet, or illness, fungi overgrow and disrupt the anti-tumour immune activity that bacteria normally support. This balance is now considered a critical regulator of cancer treatment response. Impaired CD8 T cell activity – The Cedars-Sinai study found that fungal overgrowth impairs the activity of cytotoxic CD8 T cells (the immune cells most critical for killing cancer cells) while increasing immunosuppressive macrophages in the tumour microenvironment. A Fast-Moving Field: Research Milestones The relationship between the gut microbiome and immunotherapy is one of oncology’s fastest-growing research areas. Research published in iMeta (2024) documents the following milestones: A landmark review in Nature Reviews Drug Discovery (2025) concludes that the gut microbiome is now recognised as a key modulator of immunotherapy efficacy, with clinical trials of microbiome-based interventions actively underway. The Future of Microbiome-Driven Cancer Care Researchers highlighted in a Nature collection on the microbiome in cancer (2024) note that tumours contain both bacteria and fungi, and that their presence is proving useful in detecting cancers, categorising them, and predicting whether specific interventions will work. Potential future strategies include: A 2026 review in Gut Microbes concludes that fungi and their metabolites may enhance tumour immunogenicity and reduce therapeutic resistance, with antifungal-based therapies showing particular promise as an adjunct to immunotherapy. What This Means for Cancer Patients Cancer therapy is no longer just about targeting tumour cells. The microbial ecosystem inside the body plays a significant role in determining treatment success. As Dr. David Underhill of Cedars-Sinai stated after the landmark study: “It would be really handy to be able to characterise a patient’s microbiome, both bacterial and fungal, and determine their expected prognosis, or how they will respond to radiation therapy, chemotherapy, or immunotherapy. Then we would like to see whether it is feasible to change that microbiome to make those therapies work better.” The gut microbiome, including its fungal residents, may soon become one of oncology’s most important biomarkers and therapeutic targets. FAQs Can gut fungi actually affect cancer treatment outcomes? Yes. Research in Cancer Cell shows that gut fungi directly suppress anti-tumour immune responses after radiation therapy. Antifungal treatment restored immune activity and improved outcomes in preclinical models of breast cancer and melanoma. Why do some patients not respond to immunotherapy? Multiple factors contribute, including tumour genetics, immune profile, and gut microbiome composition. Only 20 to 40% of patients in certain groups respond to immunotherapy. Scientists believe optimising the mycobiome may help improve these rates. What is Dectin-1, and why does it matter in cancer? Dectin-1 is an immune receptor that senses fungi. Higher Dectin-1 expression in
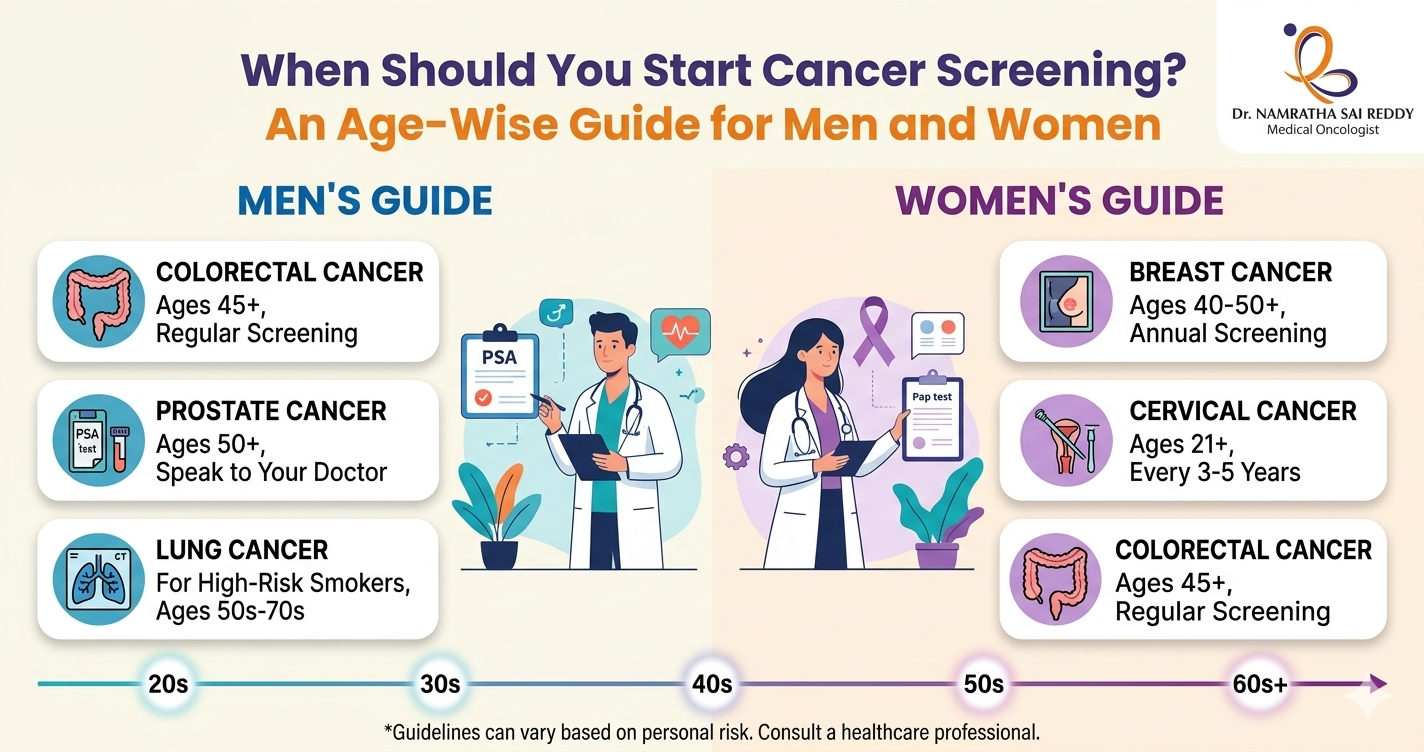
When Should You Start Cancer Screening? An Age-Wise Guide for Men and Women
• Most cancer screenings begin between the ages of 21 and 50. Missing or delaying them is one of the biggest reasons cancers go undetected until they’re harder to treat. • Women start cervical screening at 21, mammograms at 40; everyone starts colorectal screening at 45; men discuss prostate screening at 50. Knowing your age-based schedule removes the guesswork. • Pap smears and colonoscopies can catch and remove precancerous cells before they turn malignant, making them true prevention tools. • Family history, genetic mutations (BRCA, Lynch syndrome), smoking, or prior precancerous conditions can move your screening start date earlier. Talk to your doctor if any of these apply. • Regular screening, even when you feel completely fine, is what makes early detection possible. Cancer screening should begin at age 21 for women (cervical cancer) and age 45 for both men and women (colorectal cancer). Women should start mammograms at 40, and men should discuss prostate screening at 50. The right age depends on cancer type, gender, and personal risk factors. Cancer often develops silently, without obvious symptoms in its early stages. That is why screening plays a crucial role in preventive healthcare. Cancer screening tests help detect abnormal cells or early-stage cancers before symptoms appear, allowing treatment to begin when it is most effective. Why Cancer Screening Is Important Cancer screening helps detect disease before symptoms appear. Early detection significantly improves treatment outcomes and survival rates. Most early-stage cancers have survival rates of 80 to 90 percent, while advanced-stage cancers are far more difficult to treat. According to the AACR Cancer Progress Report 2024, overall cancer death rates in the United States fell by 33 percent between 1991 and 2021, translating to an estimated 4.1 million lives saved, a trend driven in large part by earlier detection through screening. Screening also identifies precancerous changes, allowing doctors to treat them before cancer develops. For example, colonoscopies and Pap tests can prevent certain cancers entirely, not just detect them early. Key benefits of cancer screening include: • Early diagnosis and timely treatment • Improved survival rates • Less aggressive treatment options • Better quality of life • Prevention of certain cancers Ages 21 to 29: Early Preventive Screening In this age group, routine screening begins primarily for women. For Women Cervical Cancer Screening Women should begin Pap smear screening at age 21, regardless of sexual activity. The recommended schedule is a Pap test every three years between the ages of 21 and 29. Pap tests detect abnormal cervical cells that may eventually develop into cervical cancer. Breast Awareness Women should begin monthly breast self-examinations from age 20 and consult a doctor if they notice lumps, breast pain, or changes in breast shape or skin. For Men Routine cancer screening is not typically recommended for most men in this age group unless there are specific risk factors. Men should still attend regular health check-ups that include general examinations for skin changes, lumps or swelling, and abnormal growths. Men with a strong family history of cancer may require earlier screening discussions with their doctor. Ages 30 to 39: Expanding Screening In your thirties, screening continues for cervical cancer and preventive health evaluations. For Women Cervical Cancer Women aged 30 to 65 should undergo either a Pap smear plus HPV test every five years, or a Pap smear alone every three years. HPV testing helps detect high-risk strains of human papillomavirus, which are responsible for approximately 99 percent of all cervical cancers. Breast Cancer Routine mammograms are not yet recommended for average-risk women in their thirties. Those with a family history, BRCA gene mutation, or other elevated risk factors should discuss earlier screening with their doctor. For Men Routine screening is still limited at this age. Men should monitor their health and consult a doctor if they notice persistent fatigue, unexplained weight loss, or lumps and unusual swelling. Men with genetic or family cancer risk may require earlier screening discussions. Ages 40 to 49: An Important Screening Stage Cancer screening becomes more structured during this decade. For Women Breast Cancer Women should begin annual mammograms starting at age 40. Mammography can detect breast cancer before symptoms appear, significantly improving survival rates. Thanks in part to early mammography, most breast cancers today are diagnosed at stage I, which carries a five-year survival rate of close to 100 percent. Cervical Cancer Continue screening with a Pap plus HPV test every five years, or a Pap test alone every three years. For Men (High Risk) Prostate Cancer Men with increased risk, such as a family history of prostate cancer or of African American descent, should discuss screening with their doctor starting at age 45. Screening may include a PSA (prostate-specific antigen) blood test and a digital rectal examination (DRE). For Both Men and Women Colorectal Cancer Routine screening for colorectal cancer should begin at age 45 for most adults. Thanks to screening, colorectal cancer death rates have fallen by 47 percent in men and 44 percent in women over recent decades. Ages 50 to 64: Comprehensive Cancer Screening In your fifties and early sixties, screening becomes essential across multiple cancer types. For Women Women should continue annual mammograms. Cervical cancer screening continues with a Pap plus HPV test every five years, or a Pap smear every three years. For Men Prostate Cancer Men aged 50 and above should discuss prostate cancer screening with their doctor. Testing may include a PSA blood test and a digital rectal exam. Shared decision-making, weighing the potential benefits and harms of screening is recommended. For Both Men and Women Colorectal Cancer Recommended screening methods include a colonoscopy every ten years, annual stool tests (FIT or FOBT), flexible sigmoidoscopy every five years, or a stool DNA test every three years. Colonoscopy remains the gold standard because it can both detect and remove precancerous polyps during the same procedure. Lung Cancer Adults aged 50 to 80 with a heavy smoking history like 20 pack-years
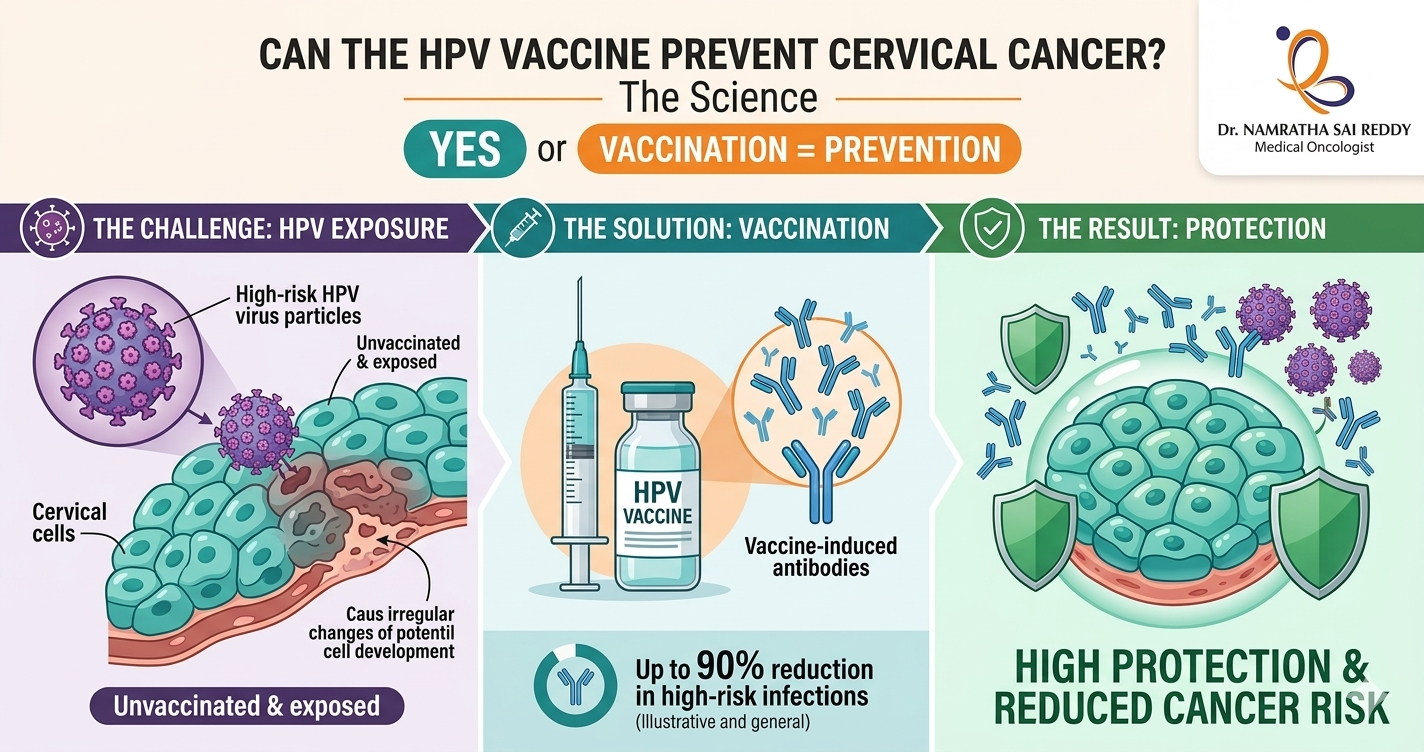
Can the HPV Vaccine Prevent Cervical Cancer? Here’s the Science
Yes, and the evidence is overwhelming. HPV vaccination reduces cervical cancer risk by up to 88–90% when given before age 17. In India, where a woman dies of cervical cancer every seven minutes, the HPV vaccine is not just a health tool; it is a national emergency intervention. India launched its nationwide free vaccination programme on 28 February 2025, targeting 11.5 million girls aged 14. Key Takeaways • India carries the world’s highest cervical cancer death toll. Over 79,906 women died from cervical cancer in India in 2022, more than 22% of all global deaths and a new case is diagnosed every four minutes. • HPV causes virtually all cervical cancers in India. HPV types 16 and 18 alone account for 76.7% of cervical cancer cases in India, making targeted vaccination extraordinarily effective. • Early vaccination delivers the strongest protection. Girls vaccinated before age 14 need only 2 doses and gain up to 90% risk reduction. Waiting until adulthood significantly reduces the vaccine’s benefit. • India now has an affordable home-grown vaccine. CERVAVAC by the Serum Institute of India costs ₹1,400–2,500 per dose in private clinics — and is free at government centres for eligible girls under the national programme. • Vaccination and screening must go together. The HPV vaccine does not protect against all high-risk strains. Regular Pap smears or HPV DNA tests remain essential, yet India’s current cervical cancer screening rate is just 2%. The Link Between HPV and Cervical Cancer Human Papillomavirus (HPV) is the most common sexually transmitted infection worldwide. While most HPV infections clear on their own, certain high-risk strains can persist and cause cancers of the cervix, vagina, vulva, penis, anus, and throat. Cervical cancer is the most common HPV-related cancer in women. According to the WHO’s Global Strategy for Cervical Cancer Elimination (2025), approximately 660,000 new cases and 350,000 deaths occurred in 2022 alone, with the highest burden in low- and middle-income countries. In India, cervical cancer claims one woman’s life every eight minutes and accounts for approximately 1.23 lakh new cases per year. Approximately 99.7% of all cervical cancers are caused by untreated or chronic HPV infection, making it one of the most preventable of all cancers. HPV types 16 and 18 alone are responsible for around 70% of all cervical cancer cases globally. How the HPV Vaccine Prevents Cervical Cancer The HPV vaccine is a preventive injection, not a treatment. It works by training your immune system to recognise specific high-risk HPV strains before exposure. When a vaccinated person later encounters the virus, their immune system neutralises it before it can establish a persistent infection that leads to cancer. Think of it as the only shot specifically designed to prevent cancer. The HPV vaccine is not just a vaccine against a virus; it is a vaccine against cancer. The “shot to prevent cervical cancer” and the “injection to prevent cervical cancer” both refer to the HPV vaccine, available in India as Gardasil, Gardasil 9, Cervarix, and CERVAVAC. It is administered as an intramuscular injection into the upper arm. HPV Vaccine Types Available in India As of 2025, India has four approved HPV vaccines. Each differs in the number of HPV strains covered, the approved age range, and cost. Gardasil 9 (Nonavalent) • Strains covered: HPV 6, 11, 16, 18, 31, 33, 45, 52, 58 (9 strains) • Age range: 9–45 years • Broadest available protection against HPV-related cancers and genital warts Gardasil 4 (Quadrivalent) • Strains covered: HPV 6, 11, 16, 18 • Age range: 9–26 years • Also prevents genital warts Cervarix (Bivalent) • Strains covered: HPV 16, 18 • Age range: 9–25 years • Targets the two main cancer-causing HPV strains CERVAVAC (Made in India – Bivalent) • Strains covered: HPV 16, 18 • Age range: 9–26 years • Developed by the Serum Institute of India, the most affordable option available • Launched in 2023 as India’s first indigenously developed HPV vaccine HPV Vaccine Dosing Schedule: 2-Dose vs 3-Dose The number of doses you need depends on your age when you receive your first shot. The younger you are when you are vaccinated, the stronger your immune response and the fewer doses you will need. 2-Dose Schedule (Ages 9–14) Children in this age group require only two doses because their immune response is stronger. 3-Dose Schedule (Ages 15 and Older) The CDC recommends routine vaccination at ages 11 or 12 (beginning as early as age 9), well before potential exposure to HPV. Immunocompromised individuals aged 9–26 require three doses regardless of their age at first vaccination. Note on single-dose schedules: As of early 2026, 89 countries, including the United States, have adopted a single-dose schedule for national immunisation programmes, particularly for Gavi-eligible low-income countries. Multi-dose schedules remain standard in India’s private market. Why Is the HPV Vaccine Not Recommended After Age 26? This is one of the most common questions about the HPV vaccine. The answer lies in biology and health economics. The CDC’s Advisory Committee on Immunisation Practices (ACIP) does not recommend routine HPV vaccination for adults over 26 for the following reasons: 1. Prior Exposure Reduces Benefit Most sexually active adults have already been exposed to one or more HPV strains by their mid-20s. Since the vaccine prevents infection but cannot treat existing HPV, its protective benefit is significantly lower in this age group. 2. Limited Cost-Effectiveness at the Population Level A landmark study by Harvard T.H. Chan School of Public Health, published in PLOS Medicine, found that vaccinating adults beyond age 26 provides limited health benefit at the population level at a substantial cost. The study modelled HPV-associated cancer and genital wart outcomes across multiple age-group scenarios. 3. FDA Approval Up
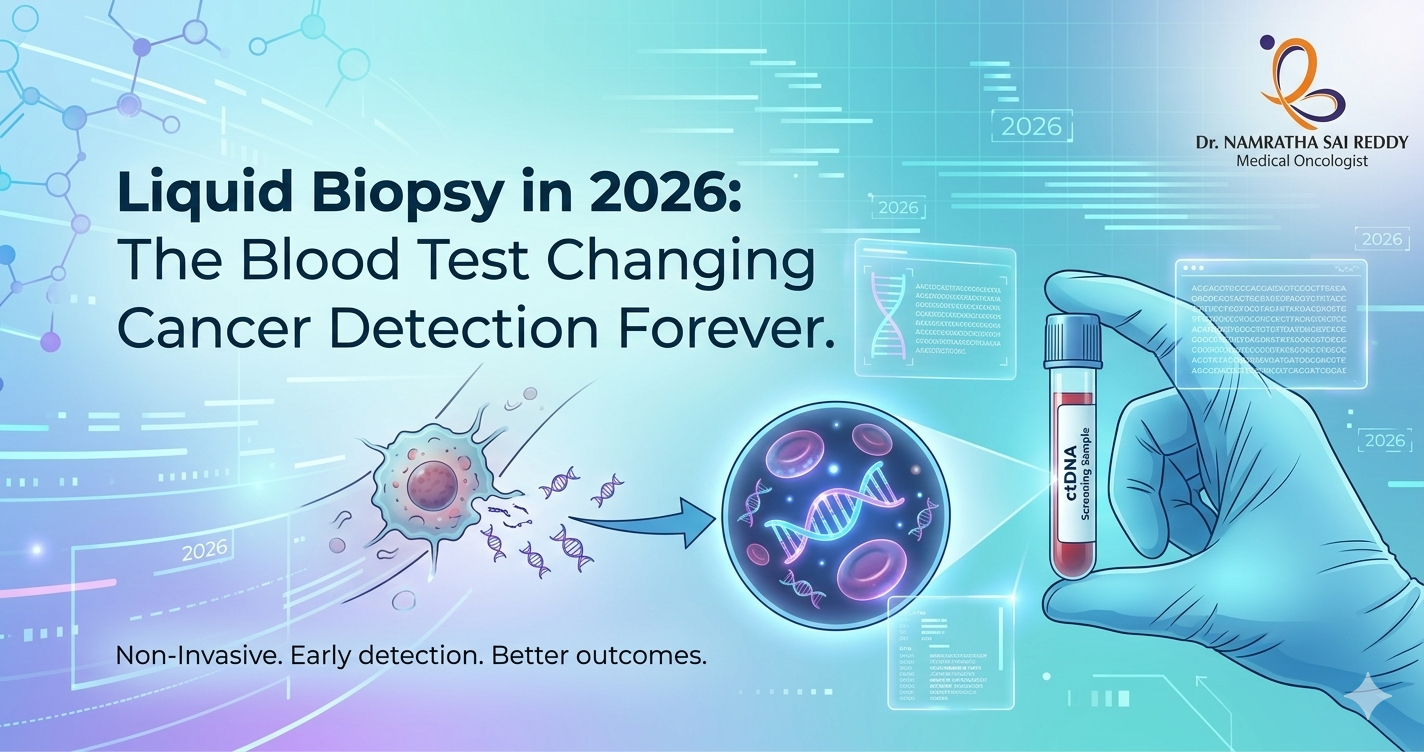
Liquid Biopsy in 2026: The Blood Test Changing Cancer Detection Forever
Liquid biopsy is a simple blood test that detects cancer DNA circulating in the bloodstream, allowing doctors to find, monitor, and track cancer without any surgery. In 2026, it has become one of the most important tools in oncology: new AI-powered tests can detect brain tumours at 98% accuracy from a blood draw alone, multi-cancer screening platforms can flag 50+ cancer types simultaneously, and a landmark U.S. law now guarantees Medicare coverage for these tests starting in 2029. For the first time, catching cancer early before symptoms appear is becoming a realistic possibility for millions of people worldwide. Key Takeaways What Is Liquid Biopsy? At its core, a liquid biopsy is any diagnostic test performed on a bodily fluid, most commonly blood, but also urine, cerebrospinal fluid, or saliva, to detect signs of cancer without physically removing tumour tissue. When cancer cells grow, divide, or die, they release biological material into the bloodstream. The most studied of these is circulating tumour DNA (ctDNA), fragments of DNA carrying the tumour’s unique genetic fingerprint. But liquid biopsies in 2026 are increasingly multimodal, analysing a rich combination of signals: When these signals are fed into machine learning models trained on millions of samples, the result is a diagnostic picture that no single tissue biopsy can match. How It Differs from Traditional Biopsy Feature Traditional Tissue Biopsy Liquid Biopsy Invasiveness Surgical procedure required Simple blood or fluid draw Tumor representation Single tumour region sampled Captures signals from tumours throughout the body Turnaround time Days to weeks Often 24–72 hours Repeatability Limited, risky to repeat often Easily repeated at regular intervals Real-time monitoring Not feasible Ideal for ongoing treatment monitoring Early detection potential Low, requires a visible tumour High, detects ctDNA before symptoms appear Risk to the patient Bleeding, infection, procedural risk Minimal — equivalent to a blood test Tumor heterogeneity Misses mutations in other regions Samples DNA from across the tumour landscape Cost (2026) $1,500–$5,000+ $300–$1,500 depending on test type The two approaches are most powerful when used together, tissue biopsy for initial diagnosis and molecular characterisation, liquid biopsy for monitoring, treatment selection, and early recurrence detection. The goal is not replacement but integration. The Science: Why ctDNA Is Such a Powerful Signal Every tumour is genetically distinct. As cancer cells proliferate and die, they leave behind fragments of DNA in the bloodstream, some as short as 160 base pairs. These fragments carry mutations specific to the tumour: point mutations, insertions, deletions, copy number variations, and methylation changes. Detecting them requires extreme precision, because in early-stage cancer, ctDNA can represent as little as 0.01% of all circulating DNA in the blood. To isolate this signal from the noise, modern liquid biopsy platforms use: The sensitivity challenge remains the field’s central technical problem, particularly for stage I cancers where tumour shedding is minimal. But every major advance in 2026 has pushed this boundary further. The Major Breakthroughs of Early 2026 1. AI + Nanosensors Solve the Brain Tumour Problem Brain tumours have historically been liquid biopsies’ most stubborn challenge. The blood-brain barrier – the tight cellular seal that protects the brain from pathogens — also blocks most tumour DNA from leaking into the bloodstream, making standard ctDNA detection unreliable for gliomas and other CNS cancers. In January 2026, Memorial Sloan Kettering Cancer Centre announced a landmark solution. Their team developed a liquid biopsy platform combining carbon nanotube sensors with artificial intelligence that detects and classifies brain tumours with 98% accuracy. Rather than searching for tumour DNA in the blood, the nanosensors detect subtle systemic changes, shifts in immune proteins, metabolic signals, and inflammatory markers throughout the body triggered by the presence of a brain tumour. Researchers describe this as reading the tumour’s “ecosystem effect” on the entire body, rather than trying to catch the few DNA fragments that escape the blood-brain barrier. Why it matters: 2. M-PACT: AI Classifies Pediatric Brain Tumours from Spinal Fluid Published in Nature Cancer in February 2026, St. Jude Children’s Research Hospital’s M-PACT (Methylation-based Predictive Algorithm for CNS Tumours) represents a parallel revolution for pediatric oncology. The algorithm analyses ctDNA extracted from cerebrospinal fluid (CSF) and uses DNA methylation patterns to classify brain tumour subtypes with 92% accuracy. Key capabilities of M-PACT include: For context, pediatric brain tumours are the leading cause of cancer-related death in children under 15. The ability to non-invasively monitor these children throughout treatment and catch relapse earlier could have a direct impact on survival rates. 3. Bridge Capture: Finally Solving the Speed-Sensitivity-Cost Triangle For years, liquid biopsy developers faced an iron triangle: you could have a test that was sensitive, fast, or cheap, but rarely all three simultaneously. High sensitivity required deep sequencing, which was slow and expensive. Simplified workflows sacrificed rare-mutation detection. In January 2026, researchers from Genomill Health, the University of Turku, and Turku University Hospital published results on Bridge Capture, a targeted next-generation sequencing enrichment method designed to break this tradeoff. Key findings: Bridge Capture is particularly significant not because it is the most sensitive test ever created, but because it may be the first to simultaneously meet the sensitivity, cost, throughput, and standardisation requirements needed for population-scale cancer screening. 4. Lung Cancer: Detecting Mutations Six Months Before Diagnosis Research published in Molecular Oncology in early 2026 from Hospital de Amor de Barretos in Brazil delivered one of the year’s most striking findings: in an asymptomatic study participant, a liquid biopsy identified a TP53 gene mutation six months before their lung cancer diagnosis. The study evaluated commercial multigene liquid biopsy panels in non-small cell lung cancer patients and found: Lung cancer kills more people globally than any other cancer, largely because most cases are caught at stage III or IV when treatment options are limited. The ability to detect it months before symptoms appear could shift the diagnosis distribution dramatically toward earlier, more treatable stages. Key Statistics at a Glance (2026) Metric Figure Brain tumour detection accuracy (AI + nanosensor) 98% Pediatric brain tumour classification accuracy (M-PACT) 92% Relapse detection lead
Vaccines for Cancer Survivors: What You Need to Know in 2026
Cancer survivors require both standard preventive vaccines, such as flu, COVID-19, shingles, pneumonia, RSV, and Hepatitis B, to protect their weakened immune systems. In addition, emerging therapeutic cancer vaccines offer promising new options to reduce the risk of cancer recurrence. Because vaccine timing and eligibility vary by individual, cancer survivors should always consult their oncologist before receiving any vaccination. Key Takeaways: Cancer survivors have 2–4× higher risk of severe infections due to weakened immunity. The influenza vaccine reduces hospitalisation risk by 40–60% in immunocompromised individuals. Cancer survivors have a 2–3× higher risk of shingles compared to the general population. Pneumococcal vaccination prevents up to 75% of invasive pneumococcal disease in high-risk adults. Personalised mRNA cancer vaccine (mRNA-4157) reduced melanoma recurrence risk by 44% in clinical trials. More than 120 cancer vaccine clinical trials are active globally, targeting multiple cancer types. What vaccines should cancer survivors get? COVID-19, flu, pneumonia, shingles (Shingrix), RSV, and Hepatitis B, as recommended by the CDC and major oncology organisations. Cancer treatments can suppress immune function for months or even years after therapy ends. In some cases, survivors may lose immunity previously gained from childhood or adult vaccinations. As a result, cancer survivors face an increased risk of severe complications from infections such as influenza, pneumonia, and COVID-19. Vaccination remains one of the most effective strategies to reduce infection-related hospitalisations, complications, and mortality in cancer survivors. Recommended vaccines for cancer survivors commonly include: Your oncologist will determine which vaccines are appropriate based on your treatment history, immune status, and age. Recommended Preventive Vaccines for Cancer Survivors 1. COVID-19 Vaccine COVID-19 remains a serious risk for immunocompromised individuals, including cancer survivors. The updated COVID-19 vaccines are safe and strongly recommended. There is no scientific evidence linking mRNA vaccines to cancer development or recurrence. Patients with weakened immunity may require booster doses every six months. Your oncologist will recommend the optimal schedule. 2. Influenza (Flu) Vaccine Cancer survivors should receive the inactivated flu vaccine annually. Influenza can lead to severe complications, including pneumonia and sepsis, in individuals with weakened immunity. The nasal spray flu vaccine, which contains live virus, is generally not recommended for immunocompromised patients. 3. Pneumococcal Vaccine (Pneumonia) Pneumococcal vaccines protect against serious infections such as pneumonia, meningitis, and bloodstream infections. Survivors who have undergone chemotherapy, splenectomy, or stem cell transplantation are particularly vulnerable and should remain up to date with pneumococcal vaccination. 4. Shingles Vaccine (Shingrix) Shingrix is a non-live vaccine recommended for cancer survivors, especially those aged 50 and above or those with weakened immunity. Shingles can cause severe nerve pain and complications, and cancer survivors are at higher risk. 5. RSV Vaccine Respiratory Syncytial Virus (RSV) can cause serious respiratory illness in older adults and immunocompromised individuals. Newly approved RSV vaccines provide protection for eligible cancer survivors, particularly those with lung involvement or weakened immunity. 6. Hepatitis B Vaccine Cancer survivors who have received blood transfusions, stem cell transplants, or immunosuppressive therapy may require Hepatitis B vaccination or boosters. Blood tests can determine whether additional doses are necessary. Important Vaccination Guidelines for Cancer Survivors Cancer survivors should follow these key vaccination principles: Individualised vaccination planning is essential for safety and effectiveness. What is a therapeutic cancer vaccine? A therapeutic cancer vaccine is a treatment that teaches the immune system to recognise cancer-specific proteins (called neoantigens) and attack any remaining or returning tumour cells. Unlike preventive vaccines, they are given after a cancer diagnosis or surgery. Personalised mRNA Cancer Vaccines Personalised mRNA vaccines are tailored to each patient’s tumour profile. These vaccines help the immune system recognize tumor-specific proteins and target cancer cells. Clinical trials have shown promising results: Regulatory approval for several mRNA cancer vaccines is expected between 2026 and 2027. DNA Cancer Vaccines DNA cancer vaccines use genetic instructions to stimulate immune cells to attack cancer. Early clinical trials have demonstrated improved survival and reduced recurrence in certain cancers, including ovarian cancer. These vaccines represent an emerging area of cancer treatment. Access to Therapeutic Cancer Vaccines Currently, most therapeutic cancer vaccines are available through clinical trials. Cancer survivors interested in these treatments should discuss the following with their oncologist: Clinical trials provide access to the most advanced cancer vaccine therapies. Vaccination Is Essential for Long-Term Cancer Survivorship Cancer survivorship requires ongoing preventive care, monitoring, and immune protection. Vaccines play a vital role in protecting survivors from serious infections and supporting long-term recovery. Preventive vaccines reduce infection risk, while therapeutic cancer vaccines offer new hope for preventing cancer recurrence. Cancer survivors should review their vaccination history with their oncologist and develop a personalised vaccination plan based on their cancer type, treatment history, and immune recovery. Vaccination is a powerful tool in protecting health and supporting long-term remission. FAQs Can cancer survivors get the COVID-19 vaccine?Yes. COVID-19 vaccines are safe and recommended. Immunocompromised survivors may require additional boosters. Which vaccines should cancer survivors avoid?Live vaccines such as FluMist, MMR, varicella, and yellow fever should be avoided during active treatment or weakened immunity. Do cancer vaccines already exist?Yes. Preventive vaccines like HPV and therapeutic vaccines such as Provenge exist. Personalised cancer vaccines are in advanced clinical trials. When can survivors get vaccinated after chemotherapy?Vaccination is usually recommended 3–6 months after chemotherapy, depending on immune recovery. Are cancer vaccines covered by insurance?Most preventive vaccines are covered. Clinical trial vaccines are typically free, while future coverage will depend on insurance policies.
Are Your Heartburn Tablets Secretly Undermining Your Cancer Treatment?
Yes, common heartburn tablets called proton pump inhibitors (PPIs) can significantly reduce the effectiveness of cancer immunotherapy and targeted drugs, and multiple large clinical studies show PPI users face up to a 37% worse survival outcome compared to cancer patients who do not take them. Key Takeaways: Every day, millions of people reach for a proton pump inhibitor (PPI) without a second thought. Omeprazole, lansoprazole, pantoprazole, and esomeprazole. These tablets are so routine that they sit on supermarket shelves next to vitamins. In cancer care, they are handed out almost reflexively to manage the stomach side effects of chemotherapy and targeted drugs. But a rapidly growing body of clinical evidence is asking a deeply uncomfortable question: could gas tablets be quietly sabotaging the cancer treatments they are prescribed alongside? The answer, according to multiple large peer-reviewed studies, is yes, and the effect is far from trivial. What Are PPIs and How Common Are They in Cancer Patients? Proton pump inhibitors work by blocking acid-producing pumps in the stomach lining, reducing gastric acid output by up to 99% at standard doses. They are prescribed to patients to control heartburn, acid reflux, nausea, and gastrointestinal side effects of immunotherapy. The most widely used include: • Omeprazole (Prilosec / Losec) • Lansoprazole (Prevacid) • Pantoprazole (Protonix) • Esomeprazole (Nexium) • Rabeprazole (Aciphex) Many of these are available without a prescription, which is precisely why they so often go unmentioned in oncology consultations. Patients simply do not realise that a heartburn tablet bought at a pharmacy could interact with their cancer drug. The scale of use is significant. Research confirms that approximately 30% of all cancer patients take PPIs, often for extended periods and frequently during active treatment windows. Some studies put the figure even higher:a large US study drawing on SEER-Medicare data found acid-reducing agent use as high as 55% across all cancer types in national databases. How PPIs Weaken Cancer Immunotherapy Immune checkpoint inhibitors (ICIs), drugs like pembrolizumab (Keytruda), nivolumab (Opdivo), and atezolizumab (Tecentriq) are among the most significant advances in cancer treatment in decades. They work by releasing the immune system’s natural brakes, allowing T-cells to identify and destroy tumour cells. But this process depends critically on a healthy, diverse gut microbiome to prime and sustain that immune response. PPIs weaken the gut microbiome. By eliminating stomach acid, they allow bacteria that would normally be killed in the stomach to colonise the gut. This suppresses beneficial strains like Bifidobacterium and Lactobacillus that are strongly associated with positive immunotherapy outcomes. The result is a blunted immune response at the exact moment treatment is trying to harness it. The clinical data make for sobering reading: • A 2023 meta-analysis published in Frontiers in Immunology analysed cancer patients receiving immune checkpoint inhibitors and found PPI users had a hazard ratio of 1.37 for overall survival (95% CI 1.23-1.52) and 1.28 for progression-free survival (95% CI 1.15-1.42), meaning they were 37% more likely to die and 28% more likely to see their cancer progress. • A 2025 systematic review and meta-analysis published in PubMed, drawing on 10,420 individuals, found PPI use linked to an 18% increased mortality risk (HR = 1.18; 95% CI: 1.11-1.25) and a 12% higher risk of disease progression in solid tumour patients receiving ICIs. • A meta-analysis in the Journal of Personalised Medicine focused on 1,015 bladder cancer patients receiving immunotherapy and found a hazard ratio of 1.55 for overall survival and 1.43 for progression-free survival, a 55% worse survival rate in PPI users. • Researchers at Strasbourg University Hospital confirmed the association held not just in lung and bladder cancer, but across multiple tumour types, and across both anti-PD-1 and anti-PD-L1 agents. Crucially, patients in control arms not receiving immunotherapy did not show the same association, strongly implicating the microbiome mechanism specifically. A November 2023 study, published in PubMed, put it plainly: “PPI users showed significantly lower progression-free survival and overall survival in the chronic use, recent use, and concomitant use groups.” The timing of PPI use relative to treatment also matters: PPI use during the first 30 days of immunotherapy appears to be particularly damaging, coinciding with the critical window in which the immune system is being activated. Not All Acid Suppressants Are Equally Harmful One of the most clinically useful findings in recent research is that histamine-2 receptor antagonists (H2RAs), a different and milder class of acid suppressant including famotidine (Pepcid), do not appear to cause the same degree of harm. In bladder cancer studies, H2RA use was not associated with worse survival outcomes, unlike PPIs. In TKI studies, H2RAs raise stomach pH less dramatically and for a shorter duration, which means drug absorption is less severely impacted. Where a PPI cannot be avoided, pharmacokinetic research suggests that taking the TKI two hours before the PPI can help preserve absorption by exploiting the pre-dose window of lower pH. Some studies have also shown that consuming the TKI with an acidic drink such as cola can enhance erlotinib bioavailability by approximately 40% in patients simultaneously taking esomeprazole. These are strategies to explore with your oncologist, not to self-manage. Long-Term PPI Use and Cancer Risk The concern does not stop at treatment interference. Long-term PPI use has itself been associated with increased cancer risk in observational research. A meta-analysis of multiple observational studies found that PPI use exceeding three months was significantly associated with elevated cancer risk through the following mechanisms: • Gut microbiome disruption, reducing immune surveillance of early cancer cells • Malabsorption of Vitamin B12, calcium, magnesium, and iron, all linked to increased cancer risk at chronically low levels • Hypergastrinemia: elevated gastrin levels caused by long-term acid suppression, which can stimulate abnormal cell proliferation in the stomach lining • Multiple studies report a dose-duration relationship for gastric
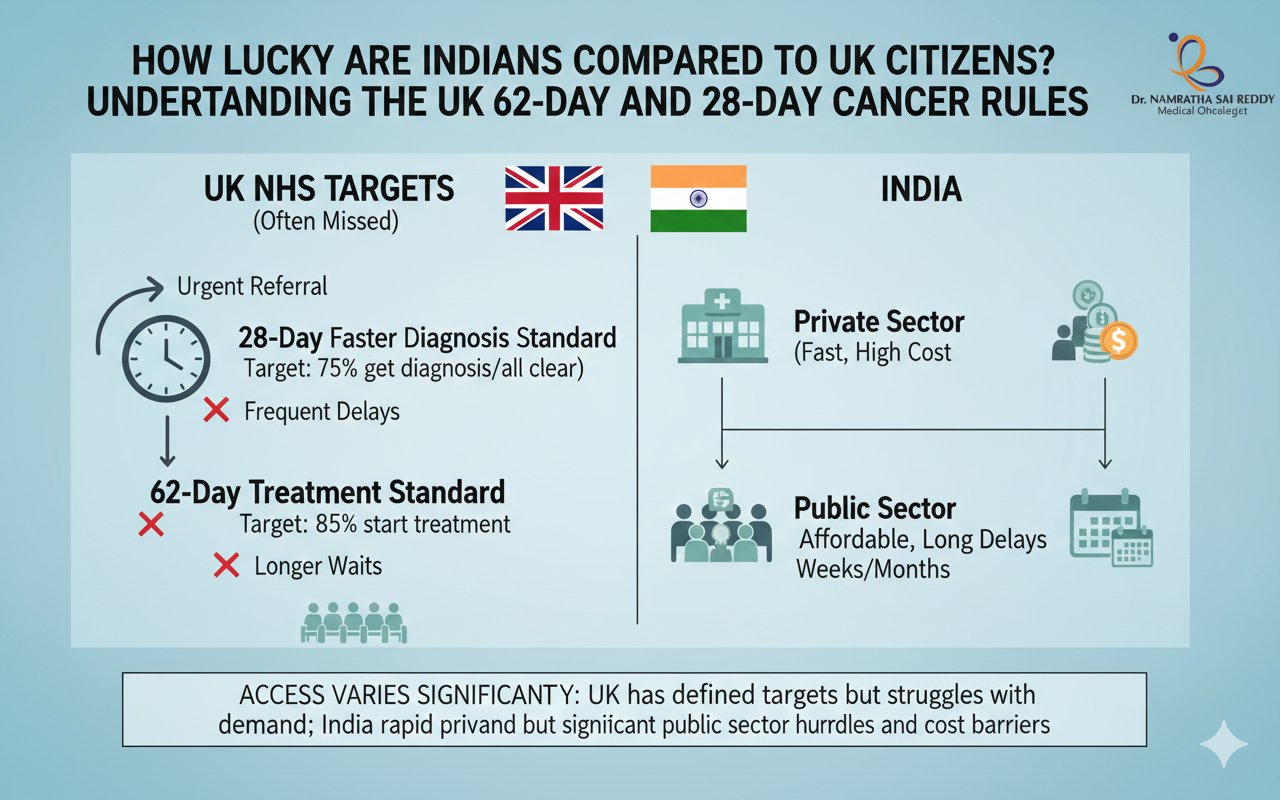
How Lucky Are Indians Compared to UK Citizens? Understanding the UK’s 62-Day and 28-Day Cancer Rules
Cancer treatment in India vs UK reveals a striking difference in how quickly patients receive life-saving care. While the UK’s NHS operates under targets like the 28-day diagnosis rule and the 62-day treatment rule, many patients still face long waiting times. In contrast, cancer treatment in India vs UK comparisons show that treatment in India’s private healthcare system often begins within a median of about 20 days, sometimes even within a week in leading metropolitan hospitals. Cancer Treatment in India vs UK: Why Treatment Starts Faster in India Indians with access to quality healthcare are significantly more fortunate than UK citizens when it comes to cancer treatment speed. While the UK’s National Health Service mandates a 28-day diagnosis and 62-day treatment start timeline (which only 70% of patients actually meet), India’s private healthcare system delivers cancer treatment within a median of 20 days, often within just 7 days in top metropolitan hospitals. Our patients have the time to take 2-3 opinions, while patients in the UK wait to start their treatment during that time. India’s $8.71 billion medical tourism industry, treating 7.3 million international patients annually, demonstrates the superior speed, affordability, and quality of care available in Indian cancer centres. Key Takeaways What Are the UK’s Cancer Waiting Time Rules? The UK National Health Service established three cancer waiting time standards in October 2023 to ensure timely diagnosis and treatment: The 28-Day Faster Diagnosis Standard Patients must receive a definitive cancer diagnosis or have cancer ruled out within 28 days of an urgent suspected cancer referral. Target: 75% of patients (increasing to 80% by March 2026). The 62-Day Referral to Treatment Standard Patients must begin cancer treatment within 62 days from when hospitals receive urgent suspected cancer referrals from GPs, screening programs, or consultant upgrades. Target: 85% of patients. The 31-Day Decision to Treat Standard Patients should start treatment within 31 days of diagnosis and the decision to treat. Target: 96% of patients. The Harsh Reality: UK NHS Failing Its Cancer Patients The UK’s cancer waiting time standards exist on paper, but the reality tells a starkly different story: Current UK Performance (November 2025) UK’s Decade-Long Failure The 62-day cancer treatment target has not been met since December 2015, that’s nearly a decade of continuous failure. The 18-week treatment target for elective procedures has not been met since 2016. The Human Cost The NHS waiting list stood at 7.31 million cases in November 2025, with approximately 154,000 patients waiting over a year for treatment. Real UK patients report: India’s Cancer Healthcare Revolution: Speed, Quality, and Affordability Lightning-Fast Treatment Timelines India’s private healthcare system operates at a speed UK patients can only dream of: Median Time to Treatment: 20 days from diagnosis, with patients receiving treatment within 7-39 days in private facilities Top Hospital Performance: Major metropolitan hospitals in Delhi, Mumbai, Chennai, Bangalore, and Hyderabad often begin treatment within days of diagnosis. Government Initiative Impact: Patients enrolled under PM-JAY health insurance saw 90% improvement in timely treatment access, and those diagnosed after 2018 had 36% higher odds of getting treatment within 30 days World-Class Success Rates Indian cancer centres deliver outcomes matching or exceeding global standards: Cutting-Edge Technology India’s top cancer centres offer technology rivalling the best facilities worldwide: Advanced Treatment Options: India: The Global Cancer Treatment Destination Massive Medical Tourism Growth India’s medical tourism market was valued at $7.69 billion in 2024, with around 7.3 million foreign patients coming to the country for treatment. This is expected to reach $8.71 billion in 2025 and $58 billion by 2035. Cancer Treatment Leadership: Oncology is advancing at a 16.73% CAGR through 2031, led by proton therapy and CAR-T cell platforms Why Patients Choose India Over the UK Study of 2,835 International Cancer Patients (55 countries, November 2013 – April 2019): The two most prominent reasons for overseas treatment were unreliable medical service at home or in nearby countries and the non-availability of medical services in the home country Key Factors: Remarkable Cost Advantage India’s medical tourism market beneficiaries pay 60-80% less than OECD rates, with bypass grafts starting at USD 5,000 against USD 100,000 in the United States Cancer Treatment Cost Comparison: India’s Quality Assurance: International Standards Government Support Infrastructure Streamlined Access: How Indians Compare to UK Citizens: The Verdict If You’re in Metropolitan India: Significantly More Fortunate Advantages: If You Have Health Insurance or Can Afford Private Care You have access to cancer treatment that is: The UK Advantage: Universal Coverage (But At What Cost?) The UK’s NHS provides universal access regardless of ability to pay, a significant advantage. However, this comes with: Why Treatment Delays Kill: The Critical Importance of Speed Delays of more than 3 months in breast cancer can decrease survival by 10-20%, while delays over 60 days in head and neck cancers can reduce 5-year survival by almost 30% The Math is Brutal: India’s Challenges: Room for Improvement While India’s private healthcare excels, challenges remain: Geographic Disparity Rural areas still lack adequate cancer screening and treatment facilities, requiring patients to travel to metropolitan centres. Public Healthcare Capacity Government hospitals face capacity constraints, though PM-JAY insurance is bridging this gap. Awareness and Early Detection A hospital-based study from northern India showed that 90% of patients with lung cancer were diagnosed at an advanced stage Ongoing Improvements: Source: Cancer Statistics 2020 Report – JCO Global Oncology What This Means for Patients and Policymakers India’s cancer care advantage lies in action, not aspiration. While countries like the UK have well-defined waiting-time targets, persistent delays continue to impact real-world outcomes. India, on the other hand, is delivering faster treatment initiation, globally comparable technology, strong survival outcomes, and affordable access at scale. For patients, this means earlier treatment and more choice. For policymakers and health systems, it reinforces a critical lesson: speed, access, and execution matter as much as guidelines. India’s model shows that timely, high-quality cancer care is achievable when systems are designed around outcomes, not just targets. FAQs 1. What is the UK 62-day cancer rule?It is an NHS
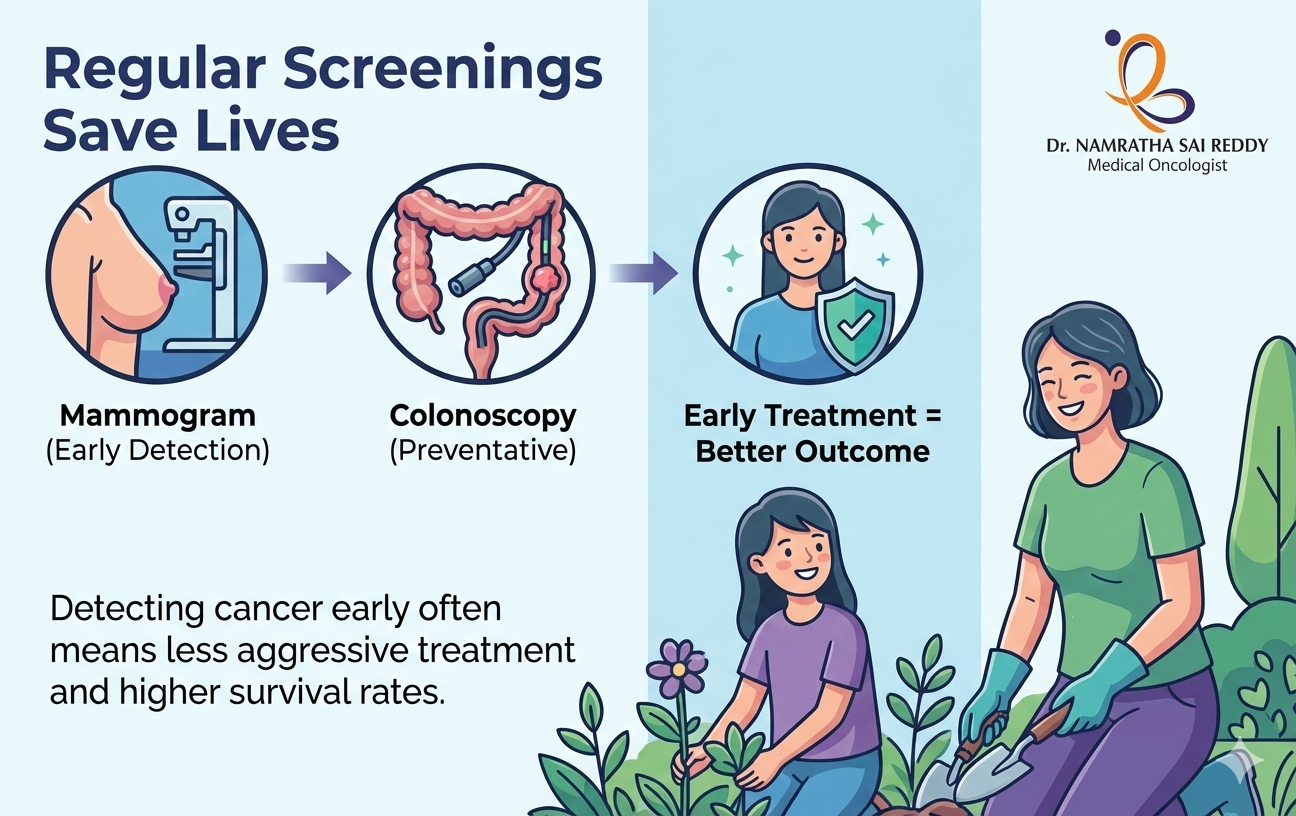
How Regular Screenings Save Lives: Mammograms, Colonoscopy, and More
Cancer rarely begins with pain or visible warning signs. In many people, it develops silently over the years, spreading before symptoms appear. This quiet progression is exactly why preventive cancer screening is one of the most powerful tools in modern healthcare. Regular screening allows doctors to detect cancer early or stop it before it starts. It plays a central role in cancer screening and prevention, reducing deaths and improving long‑term outcomes. When screening becomes routine, cancer shifts from a crisis to a condition that can often be managed or cured. Cancer Screening: What It Really Means Cancer screening refers to medical tests performed on people who feel healthy and have no symptoms. The purpose of a screening examination is to identify early cancer, precancerous changes, or risk indicators that need closer follow‑up. These tests are designed to raise an alert, not to deliver a final diagnosis. Screening works because cancer usually develops in stages. Early cellular changes may not cause symptoms, but they can still be detected through imaging, lab tests, or physical exams. This is the foundation of screening and early detection of cancer. It is important to understand that screening is not perfect. Some tests may miss disease, while others may flag harmless changes. Still, when applied correctly and repeated on schedule, screening dramatically improves survival and supports the primary prevention of cancer. Key goals of cancer screening include: Cancer Screening and Prevention: How Screening Stops Cancer Before It Starts Many people assume screening only finds cancer. In reality, it often prevents cancer entirely. Several cancers begin as abnormal but non‑cancerous changes that can be treated or removed early. This is where cancer screening and prevention intersect. For example, a colonoscopy removes polyps before they turn cancerous. Cervical screening detects abnormal cells long before cancer develops. These actions represent true primary prevention of cancer. Screening also opens the door to risk reduction. When tests reveal elevated risk, doctors can guide lifestyle changes, monitor closely, or begin preventive treatment. This proactive approach lowers future cancer burden at both the individual and population levels. How screening contributes to prevention: Why Early Detection of Cancer Saves Lives Cancer outcomes depend heavily on stage at diagnosis. Early‑stage cancers are usually localised, smaller, and easier to treat. This makes early detection of cancer the single most important factor in survival. An early cancer detection test often allows simpler treatment. Surgery may replace chemotherapy. Recovery is faster, and long‑term side effects are reduced. Patients also experience less emotional and financial strain. This is why public health systems prioritise screening and early detection of cancer. The earlier cancer is found, the greater the chance of a cure and long‑term survival. Benefits of early detection include: When Should You Get Screened? There is no single age that fits everyone. Screening schedules depend on age, gender, family history, and lifestyle risks. A personalised cancer check-up test plan is always more effective than a generic one. Some screenings begin in early adulthood, while others start later. High‑risk individuals may need earlier or more frequent tests. Your healthcare provider evaluates these factors to recommend the right screening exam at the right time. Factors that influence screening timing: Screening for Common Cancer Breast Cancer Screening (Mammograms) Mammograms are one of the most proven cancer screening tools. They detect breast cancer before lumps can be felt, often years earlier. This makes them essential among cancer screening tests for females. Regular mammography significantly lowers breast cancer mortality. Early detection allows for breast‑conserving treatment and improved survival. For women at higher risk, additional imaging may be recommended. Cervical Cancer Screening Cervical screening does more than detect cancer. It prevents it. Pap smears and HPV tests identify abnormal cells that may later become cancerous. This form of screening represents one of the strongest examples of primary prevention of cancer. When done regularly, cervical cancer becomes largely preventable. Colon Cancer Screening Colon cancer screening both detects and prevents disease. Colonoscopy allows doctors to remove polyps before they turn malignant. Other methods, such as stool‑based tests, also play a role in screening for common cancers, especially when a colonoscopy is not feasible. Other Essential Cancer Screening Tests Lung Cancer Screening Low-dose CT scans are recommended for high-risk individuals with a smoking history. These scans support early detection of cancer when lung cancer is still treatable. Prostate Cancer Screening PSA blood tests may help detect prostate cancer early in select age groups, depending on risk. Skin Cancer Screening Full-body skin exams help identify melanoma and other skin cancers early, even before symptoms appear. Together, these form a comprehensive approach to screening for common cancers across populations. Can We Detect Cancer Through Blood Tests? Blood tests can sometimes detect cancer markers or genetic changes. They may also help monitor existing cancer. New multi‑cancer blood tests show promise, but they are not replacements for established screening. At present, they complement rather than replace standard cancer detection methods. Blood tests are best viewed as part of a broader cancer screening and prevention strategy, not a standalone solution. Understanding Screening Results and Follow-Up A screening result is not a diagnosis. Cancer grows slowly. That’s why repeating tests at recommended intervals is essential for sustained cancer detection. How to Detect Cancer at an Early Stage: What You Can Do If you wonder how to detect cancer at early stage, consistency matters most. Following recommended schedules is more effective than one‑time testing. Steps you can take: This approach strengthens screening and early detection of cancer over time. Questions Everyone Should Ask Their Doctor These conversations personalise your cancer screening and prevention plan. Screening Is an Investment in Life Cancer screening is not about waiting for disease. It is about acting before cancer takes control. Through preventive cancer screening, people gain time, options, and often a cure. Regular screening transforms outcomes by enabling early detection of cancer, reducing treatment intensity, and improving survival. It also strengthens long‑term cancer screening and prevention strategies at both personal and public health levels. Mammograms, colonoscopy, cervical screening,
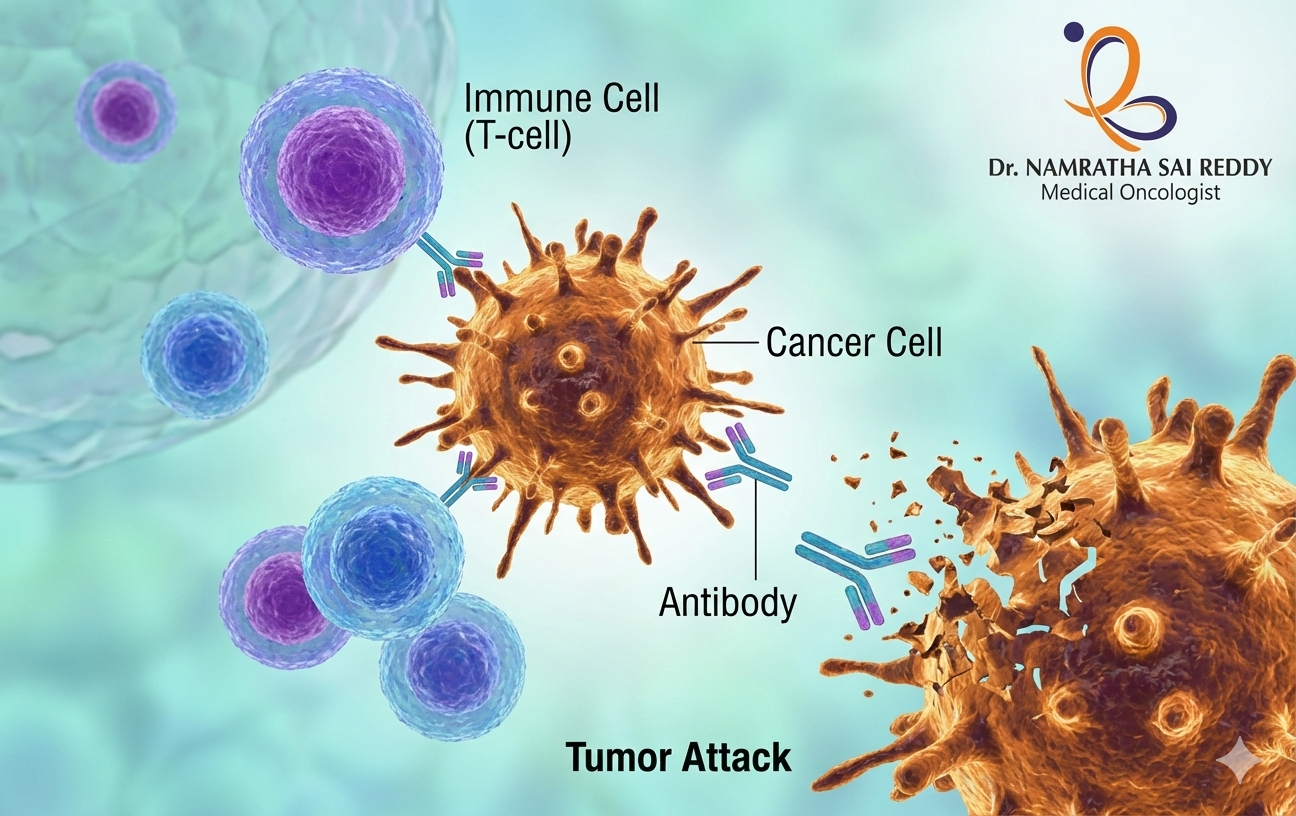
The Latest in Immunotherapy: How Cancer Treatment is Evolving
Cancer care has undergone a profound transformation in the last decade, and immunotherapy is at the centre of this change. Unlike traditional treatments that directly target tumors, immunotherapy works by strengthening the body’s own immune defenses against cancer. This shift has opened new possibilities for patients with advanced, recurrent, or treatment-resistant cancers. As research progresses, doctors are learning how to use immunotherapy drugs more precisely and safely. In this blog, we explore the immunotherapy meaning, how treatment is given, its effectiveness, costs in India, and what patients should realistically expect from this rapidly evolving approach to cancer care. What Is Immunotherapy? The immunotherapy meaning refers to treatments that help your immune system recognise and fight cancer. Your immune system is designed to detect abnormal cells, including cancer cells, and destroy them before they cause harm. Cancer becomes dangerous when it learns to hide from immune detection. Through complex signalling pathways, tumours can suppress immune responses and continue growing unchecked. Immunotherapy of cancer is treated with medicines that reverse this immune suppression. Instead of attacking cancer cells directly, immunotherapy treatment empowers immune cells to do their job more effectively. This approach often leads to more targeted cancer control with less damage to healthy tissue compared to traditional therapies. How Does Immunotherapy Work? Understanding how immunotherapy works helps explain why outcomes can vary between patients. Immunotherapy targets the interaction between cancer cells and immune cells rather than the tumour itself. Cancer cells often produce signals that tell immune cells to stop attacking. Immunotherapy cancer drugs interrupt these signals, allowing immune cells to stay active and focused on destroying cancer. Key mechanisms include: Because this process depends on your immune system, response rates differ. This variability explains why is immunotherapy effective for some patients and less effective for others. Types of Immunotherapy Used in Cancer Treatment There are several types of immunotherapy currently approved for cancer care. Each type works differently and is chosen based on cancer type, stage, and immune markers. Types of immunotherapy include: These immunotherapy drugs may be used alone or combined with other treatments such as chemotherapy or targeted therapy. Selection is always personalised to maximise benefit and reduce risk. Checkpoint Inhibitors Checkpoint inhibitors are among the most widely used immunotherapy cancer drugs today. They help immune cells called T cells remain active against cancer. Normally, checkpoint proteins prevent T cells from attacking healthy tissue. Cancer exploits these checkpoints to shut down immune responses. Immunotherapy drugs known as checkpoint inhibitors block this shutdown. By keeping T cells active, these drugs allow sustained immune attacks on tumours. Checkpoint inhibitors have shown success in cancers such as lung cancer, melanoma, kidney cancer, and some gastrointestinal cancers. Adoptive Cell Therapy (T-Cell Transfer Therapy) Adoptive cell therapy involves modifying a patient’s immune cells in a laboratory. These enhanced cells are then returned to the body to fight cancer more effectively. Main types include: Monoclonal Antibody Therapy Monoclonal antibodies are lab-engineered proteins designed to attach to specific cancer markers. Once attached, they help the immune system destroy cancer cells. Some monoclonal antibodies: These treatments are often categorised as immunotherapy drugs and targeted therapies. They play a major role in breast cancer, colorectal cancer, and lymphoma. Cancer Vaccines Cancer vaccines are different from vaccines that prevent infections. Instead, they train the immune system to recognise cancer cells already present in the body. These vaccines expose immune cells to cancer-specific markers. Once trained, immune cells can seek out and destroy cancer cells more efficiently. Cancer vaccines are still evolving, but they represent a promising area of immunotherapy treatment, especially when combined with other immune-based approaches. Immune System Modulators (Immunomodulators) Immune system modulators broadly enhance immune responses. Because they act in different ways, they are sometimes called nonspecific immune agents. Examples include: Some treatments, such as BCG for bladder cancer, are given as a localised cancer injection. These therapies have been used successfully for decades in specific cancers. How Is Immunotherapy Done? Patients often ask, how immunotherapy is done in practice. The answer depends on the specific drug and cancer type. Before starting treatment, doctors perform tests to evaluate immune markers and overall health. This ensures the chosen immunotherapy procedure is appropriate and safe. Immunotherapy may be given as: Immunotherapy Procedure: Step-by-Step Overview The immunotherapy procedure typically follows a structured plan designed to balance effectiveness and safety. Step What Happens Evaluation Cancer testing and immune profiling Planning Selection of suitable immunotherapy drugs Treatment Infusion, pill, or injection Monitoring Blood tests and imaging Cycles Treatment with scheduled breaks Treatment may be continuous or cyclical. Rest periods allow the body to recover while maintaining immune activity. Is Immunotherapy Effective? A common question looms about the effectiveness of immunotherapy. The answer is no, but its impact can be significant for the right patients. Effectiveness depends on: Some patients experience dramatic and long-lasting responses. Others may see modest benefit or none at all. Immunotherapy Success Rate for Cancer There is no single immunotherapy success rate for cancer. Success varies widely across cancer types and patient populations. For certain cancers, immunotherapy has doubled or tripled survival rates compared to older treatments. In others, it works best as part of a combination therapy. Ongoing research continues to refine patient selection to improve outcomes. Can Immunotherapy Cure Stage 4 Cancer? Many patients ask this question. In most cases, immunotherapy does not provide a complete cure. However, it can: In some patients, advanced cancer becomes a manageable chronic condition. This represents a major shift in cancer care. Immunotherapy Cost in India Cost is a major concern for patients. Immunotherapy cost in India varies based on several factors. Factor Impact on Cost Drug type Imported drugs cost more Duration Longer treatment increases cost Cancer type Combination therapy raises cost Hospital Public vs private setting Doctors help patients balance potential benefit, affordability, and safety when planning treatment. Recovery and Outlook Recovery during immunotherapy differs from traditional treatments. Many patients maintain better daily functioning compared to chemotherapy. Side effects are usually immune-related and may include fatigue, skin